PayPal, credit cards. Download editable-PDF & invoice in 1 second!
YY 1082-2007 English PDF (YY1082-2007)
YY 1082-2007 English PDF (YY1082-2007)
Precio habitual
$150.00 USD
Precio habitual
Precio de oferta
$150.00 USD
Precio unitario
/
por
Los gastos de envío se calculan en la pantalla de pago.
No se pudo cargar la disponibilidad de retiro
Delivery: 3 seconds. Download true-PDF + Invoice.
Get QUOTATION in 1-minute: Click YY 1082-2007
Historical versions: YY 1082-2007
Preview True-PDF (Reload/Scroll if blank)
YY 1082-2007: Rigid arthroscope
YY 1082-2007
YY
PHARMACEUTICAL INDUSTRY STANDARD
OF THE PEOPLE’S REPUBLIC OF CHINA
ICS 11.040.70
C 40
Replacing YY 91082-1999
Rigid arthroscope
硬性关节内窥镜
ISSUED ON: JULY 02, 2007
IMPLEMENTED ON: MARCH 01, 2008
Issued by: China Food and Drug Administration
Table of Contents
Foreword ... 3
1 Scope ... 4
2 Normative references ... 4
3 Composition ... 5
4 Requirements ... 5
5 Test methods ... 8
6 Inspection rules ... 16
7 Signs, labels and instruction manual ... 18
8 Packaging, transportation, storage ... 19
Appendix A (Normative) Safety requirements for interconnection with medical
electrical equipment ... 20
Rigid arthroscope
1 Scope
This standard specifies the classification and marking, requirements, test
methods, inspection rules, signs, labels and instruction manual, packaging,
transportation, storage of rigid arthroscopes.
This standard is applicable to rigid arthroscope (hereinafter referred to as
arthroscope). Arthroscope is suitable for the examination and diagnosis of
human joint diseases in medical clinics, as well as for treatment together with
relevant surgical systems.
2 Normative references
The provisions in following documents become the provisions of this standard
through reference in this standard. For the dated references, the subsequent
amendments (excluding corrections) or revisions do not apply to this standard;
however, parties who reach an agreement based on this standard are
encouraged to study if the latest versions of these documents are applicable.
For undated references, the latest edition of the referenced document applies.
GB/T 191-2000 Packaging - Pictorial marking for handling of goods
GB/T 2829-2002 Sampling procedures and tables for periodic inspection by
attributes (Apply to inspection of process stability)
GB/T 6463-2005 Metallic and other inorganic coatings - Review of methods
of measurement of thickness
GB 9706.1-1995 Medical electrical equipment - Part 1: General
requirements for safety (idt IEC 601-1:1988)
GB 9706.19-2000 Medical electrical equipment - Part 2: Particular
requirements for the safety of endoscopic equipment (idt IEC 60601-2-
18:1996)
GB 11244-2005 General requirements for the medical endoscope and
endoscope accessories
GB/T 14710-1993 The environmental requirements and test methods for
medical electrical equipment
flowrate of water shall be not less than 200 mL/min.
4.8 The head of the puncture needle shall be smooth and sharp, the head of
the blunt obturator shall be smooth and round, the handle of the needle of the
instrument shall be flat and smooth.
4.9 The inner hole of the instrument puncture cannula shall be clean and
smooth, which may allow the instrument puncture needle and the matching
surgical instruments to pass smoothly.
4.10 The hardness of the tip of the puncture needle and the instrument puncture
needle shall be HRC 36 ~ 46.
4.11 The welded parts of the arthroscope shall be firm and reliable, flat and
smooth, there is no insufficient welding or surfacing.
4.12 The plating of arthroscope’s plated components shall comply with the
category-V grade-2 requirements of YY 0076-1992.
4.13 Arthroscope is a medical device that is in short-term contact with the
injured surface. The outer surface material of the insertion part shall be made
of materials that have been proven to be biocompatible. Otherwise, the
following tests shall be carried out:
a) The cytotoxicity score shall be not more than 1;
b) The type of stimulus reaction shall be not more than mild;
c) There shall be no sensitization.
4.14 The safety requirements for the interconnection and use with medical
electrical equipment:
The arthroscope shall comply with the requirements of GB 9706.1-1995 and
GB 9706.19-2000. See Appendix A (Normative) for specific requirements.
4.15 The environmental test of arthroscope shall meet the requirements of
Group-2 of the climatic environmental conditions, Group-2 of mechanical
environmental conditions and Table 3 in GB/T 14710-1993.
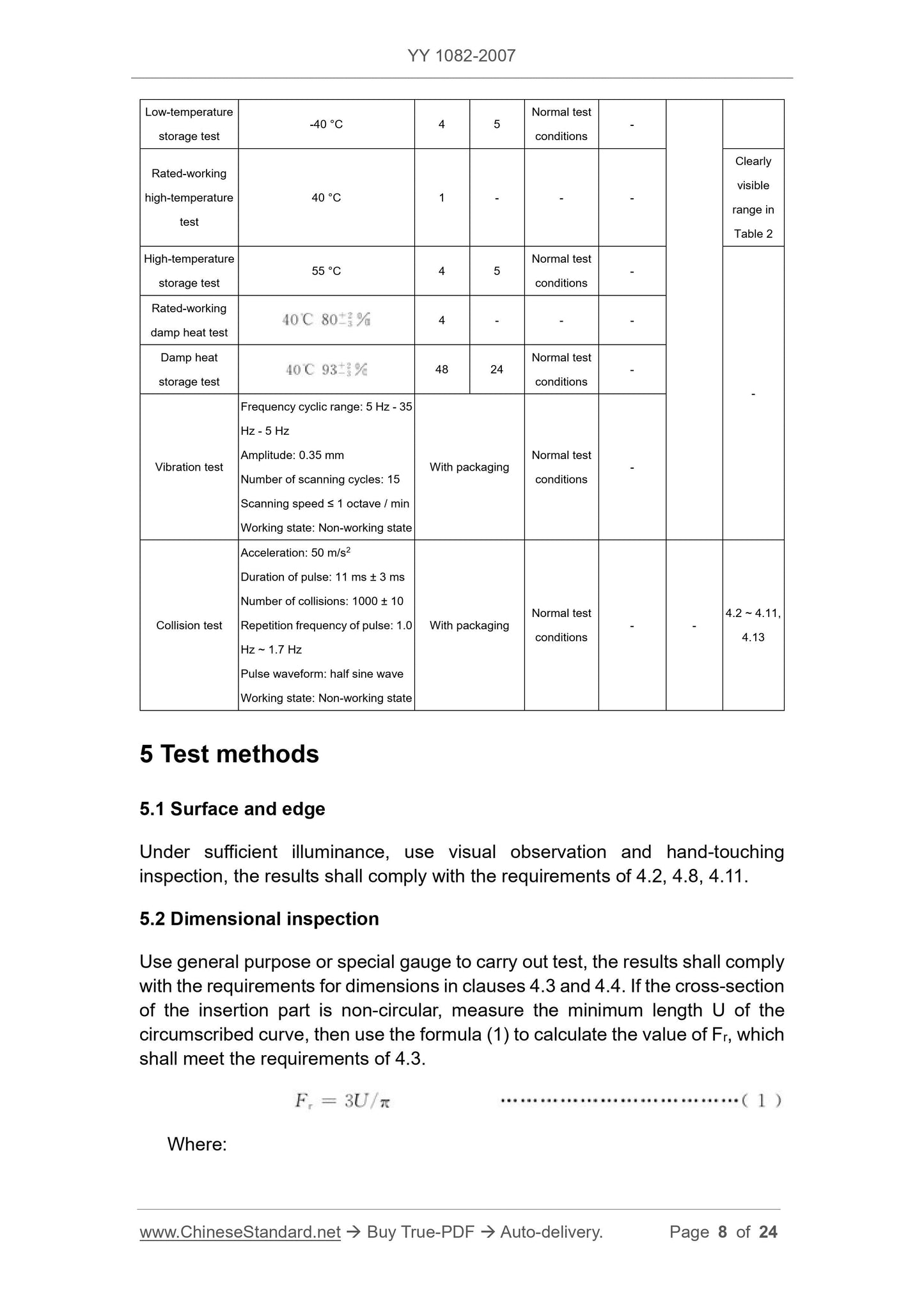
Table 3 -- Environmental test requirements and inspection items
Test item Test conditions
Test requirements Inspection items
Duration /
Recovery
time / h
Testing
environment
Initial
inspection
Middle
inspection
Final
inspection
Rated-working
low-temperature
test
5 °C 1 - -
4.2 ~ 4.11,
4.13
- -
Fr - The equivalent perimeter, in millimeters (mm);
U - The minimum length of the externally-tangent curve, in millimeters (mm).
5.3 Basic parameters of endoscope
5.3.1 Field of view and angle of view
5.3.1.1 Method-1
5.3.1.1.1 The measuring instrument consists of the following parts:
a) Optical bench or similar device, which can support the endoscope for
testing, it may adjust the optical axis of the endoscope to coincide with the
center of the measuring target. At the end surface of the endoscope’s
head, measure the field of view at a point along the perpendicular direction
50 mm from the center point of the measuring target of the concentric
circle where the angle is marked;
b) The target holder and the dial divided by “degrees” (see Figure 1);
c) Measuring target for the measurement of field of view and angle of view
(see Figure 2): circular, with a set of measurements in unit of “degree”,
measured at 50 mm. The ring of the measuring target may be calculated
according to the formula (2):
Where:
D - The diameter of the measuring ring corresponding to different angles
of view, in millimeters (mm);
β - Field of view, in degrees (°).
The measuring target is fixed on the instrument.
There shall be a main marking line every 10°, as well as the corresponding
degree indications.
A line shall be marked every 2° between each main marking line, 4
marking lines in total.
d) Lighting shall be measured by white-light-illuminated measuring target,
the minimum illuminance is not less than 500 lx, it may use an endoscope
or external light source.
5.3.1.1.2 The following test methods are only recommended:
Fix the endoscope and adjust the distance between the objective lens and the
object to be observed. When the distance is at two positions: 1 mm and 50 mm,
it may clearly observe a 1.2 mm width line through the endoscope’s eyepiece,
which shall comply with the requirements of clause 4.4.
5.4 Endoscope’s performance
5.4.1 Defect and light spot of field of view
Turn on the cold light source and adjust to the brightest state. Observe the
illuminated white paper through the endoscope, which shall meet the
requirements of 4.5.1 and 4.5.5.
5.4.2 Test of half-unshielded eyepiece cover
Test according to the method of 5.5 in GB 11244-2005, the results shall meet
the requirements of 4.5.2.
5.4.3 Test of fog layer
Insert the insertion part of the endoscope into water which has a temperature
of 20 °C or less. After 10 min, take it out. Then insert it into water which has a
temperature of 40 °C. Take it out and wipe it dry. Observe from the eyepiece, it
shall meet the requirements of 4.5.3.
5.4.4 Sealing test
Test according to the method in YY 0068-1992, the results shall meet the
requirements of 4.5.4.
5.4.5 Illuminance uniformity
Place the illuminometer probe at a distance of 50 mm in front of the endoscope.
According to the angle of view of the endoscope, adjust the illuminometer probe,
to make it perpendicular to the view direction o...
Get QUOTATION in 1-minute: Click YY 1082-2007
Historical versions: YY 1082-2007
Preview True-PDF (Reload/Scroll if blank)
YY 1082-2007: Rigid arthroscope
YY 1082-2007
YY
PHARMACEUTICAL INDUSTRY STANDARD
OF THE PEOPLE’S REPUBLIC OF CHINA
ICS 11.040.70
C 40
Replacing YY 91082-1999
Rigid arthroscope
硬性关节内窥镜
ISSUED ON: JULY 02, 2007
IMPLEMENTED ON: MARCH 01, 2008
Issued by: China Food and Drug Administration
Table of Contents
Foreword ... 3
1 Scope ... 4
2 Normative references ... 4
3 Composition ... 5
4 Requirements ... 5
5 Test methods ... 8
6 Inspection rules ... 16
7 Signs, labels and instruction manual ... 18
8 Packaging, transportation, storage ... 19
Appendix A (Normative) Safety requirements for interconnection with medical
electrical equipment ... 20
Rigid arthroscope
1 Scope
This standard specifies the classification and marking, requirements, test
methods, inspection rules, signs, labels and instruction manual, packaging,
transportation, storage of rigid arthroscopes.
This standard is applicable to rigid arthroscope (hereinafter referred to as
arthroscope). Arthroscope is suitable for the examination and diagnosis of
human joint diseases in medical clinics, as well as for treatment together with
relevant surgical systems.
2 Normative references
The provisions in following documents become the provisions of this standard
through reference in this standard. For the dated references, the subsequent
amendments (excluding corrections) or revisions do not apply to this standard;
however, parties who reach an agreement based on this standard are
encouraged to study if the latest versions of these documents are applicable.
For undated references, the latest edition of the referenced document applies.
GB/T 191-2000 Packaging - Pictorial marking for handling of goods
GB/T 2829-2002 Sampling procedures and tables for periodic inspection by
attributes (Apply to inspection of process stability)
GB/T 6463-2005 Metallic and other inorganic coatings - Review of methods
of measurement of thickness
GB 9706.1-1995 Medical electrical equipment - Part 1: General
requirements for safety (idt IEC 601-1:1988)
GB 9706.19-2000 Medical electrical equipment - Part 2: Particular
requirements for the safety of endoscopic equipment (idt IEC 60601-2-
18:1996)
GB 11244-2005 General requirements for the medical endoscope and
endoscope accessories
GB/T 14710-1993 The environmental requirements and test methods for
medical electrical equipment
flowrate of water shall be not less than 200 mL/min.
4.8 The head of the puncture needle shall be smooth and sharp, the head of
the blunt obturator shall be smooth and round, the handle of the needle of the
instrument shall be flat and smooth.
4.9 The inner hole of the instrument puncture cannula shall be clean and
smooth, which may allow the instrument puncture needle and the matching
surgical instruments to pass smoothly.
4.10 The hardness of the tip of the puncture needle and the instrument puncture
needle shall be HRC 36 ~ 46.
4.11 The welded parts of the arthroscope shall be firm and reliable, flat and
smooth, there is no insufficient welding or surfacing.
4.12 The plating of arthroscope’s plated components shall comply with the
category-V grade-2 requirements of YY 0076-1992.
4.13 Arthroscope is a medical device that is in short-term contact with the
injured surface. The outer surface material of the insertion part shall be made
of materials that have been proven to be biocompatible. Otherwise, the
following tests shall be carried out:
a) The cytotoxicity score shall be not more than 1;
b) The type of stimulus reaction shall be not more than mild;
c) There shall be no sensitization.
4.14 The safety requirements for the interconnection and use with medical
electrical equipment:
The arthroscope shall comply with the requirements of GB 9706.1-1995 and
GB 9706.19-2000. See Appendix A (Normative) for specific requirements.
4.15 The environmental test of arthroscope shall meet the requirements of
Group-2 of the climatic environmental conditions, Group-2 of mechanical
environmental conditions and Table 3 in GB/T 14710-1993.
Table 3 -- Environmental test requirements and inspection items
Test item Test conditions
Test requirements Inspection items
Duration /
Recovery
time / h
Testing
environment
Initial
inspection
Middle
inspection
Final
inspection
Rated-working
low-temperature
test
5 °C 1 - -
4.2 ~ 4.11,
4.13
- -
Fr - The equivalent perimeter, in millimeters (mm);
U - The minimum length of the externally-tangent curve, in millimeters (mm).
5.3 Basic parameters of endoscope
5.3.1 Field of view and angle of view
5.3.1.1 Method-1
5.3.1.1.1 The measuring instrument consists of the following parts:
a) Optical bench or similar device, which can support the endoscope for
testing, it may adjust the optical axis of the endoscope to coincide with the
center of the measuring target. At the end surface of the endoscope’s
head, measure the field of view at a point along the perpendicular direction
50 mm from the center point of the measuring target of the concentric
circle where the angle is marked;
b) The target holder and the dial divided by “degrees” (see Figure 1);
c) Measuring target for the measurement of field of view and angle of view
(see Figure 2): circular, with a set of measurements in unit of “degree”,
measured at 50 mm. The ring of the measuring target may be calculated
according to the formula (2):
Where:
D - The diameter of the measuring ring corresponding to different angles
of view, in millimeters (mm);
β - Field of view, in degrees (°).
The measuring target is fixed on the instrument.
There shall be a main marking line every 10°, as well as the corresponding
degree indications.
A line shall be marked every 2° between each main marking line, 4
marking lines in total.
d) Lighting shall be measured by white-light-illuminated measuring target,
the minimum illuminance is not less than 500 lx, it may use an endoscope
or external light source.
5.3.1.1.2 The following test methods are only recommended:
Fix the endoscope and adjust the distance between the objective lens and the
object to be observed. When the distance is at two positions: 1 mm and 50 mm,
it may clearly observe a 1.2 mm width line through the endoscope’s eyepiece,
which shall comply with the requirements of clause 4.4.
5.4 Endoscope’s performance
5.4.1 Defect and light spot of field of view
Turn on the cold light source and adjust to the brightest state. Observe the
illuminated white paper through the endoscope, which shall meet the
requirements of 4.5.1 and 4.5.5.
5.4.2 Test of half-unshielded eyepiece cover
Test according to the method of 5.5 in GB 11244-2005, the results shall meet
the requirements of 4.5.2.
5.4.3 Test of fog layer
Insert the insertion part of the endoscope into water which has a temperature
of 20 °C or less. After 10 min, take it out. Then insert it into water which has a
temperature of 40 °C. Take it out and wipe it dry. Observe from the eyepiece, it
shall meet the requirements of 4.5.3.
5.4.4 Sealing test
Test according to the method in YY 0068-1992, the results shall meet the
requirements of 4.5.4.
5.4.5 Illuminance uniformity
Place the illuminometer probe at a distance of 50 mm in front of the endoscope.
According to the angle of view of the endoscope, adjust the illuminometer probe,
to make it perpendicular to the view direction o...
Share